What is the Aurora?
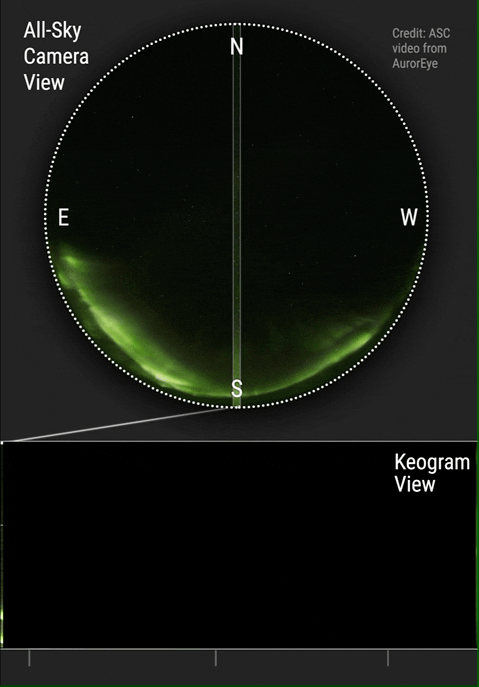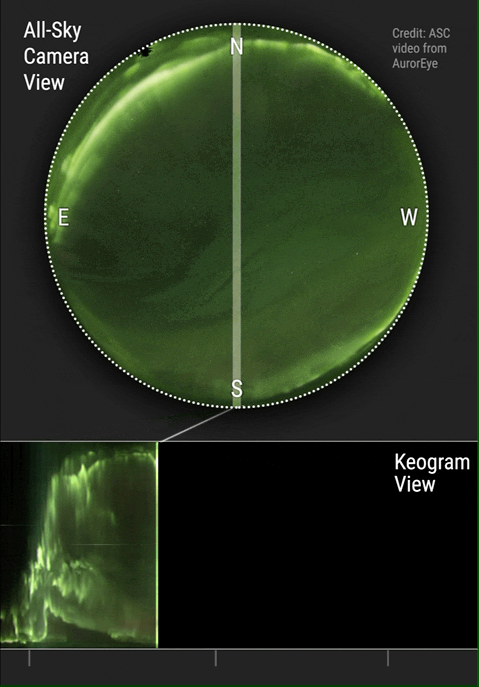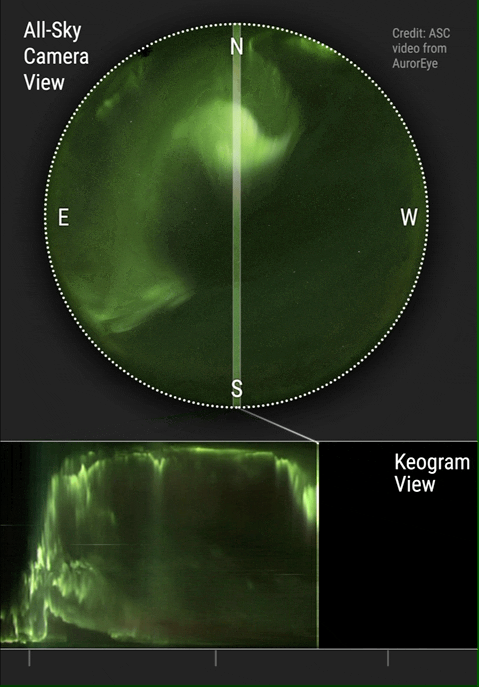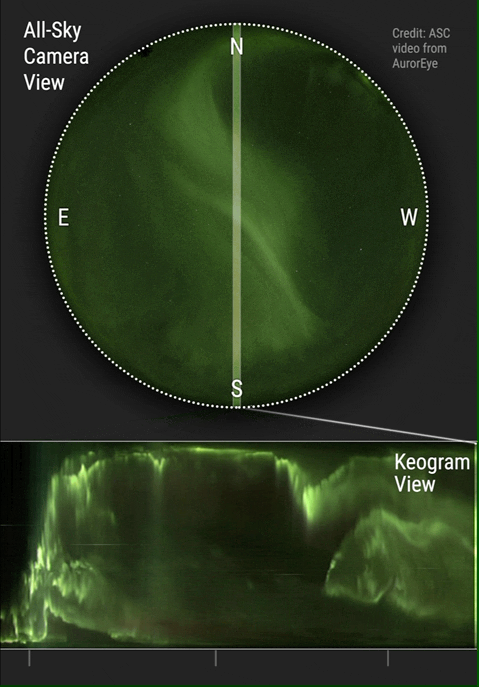
The aurora borealis or Northern Lights is an atmospheric phenomenon that occurs in the northern hemisphere of the planet. It also occurs in the southern hemisphere where it is known as the aurora australis. Aurora are known to exist on comets, brown dwarfs, other planets and on some moons. Aurora are caused by solar wind interacting with magnetic fields and atmospheric particles. Atmospheric particles become energized and form what is known as plasma. Plasma is energized or ionized gas. It is the fourth state of matter and has been so identified since about 1972. Yes, your high school teachers who taught you the three states of matter were...well...not current in their physics. You see plasma every day when you see the sun, an operating fluorescent light or an operating neon light.
Spectroscopy
Spectroscopy is the study of the interaction of matter with electromagnetic radiation. Recall that light is electromagnetic radiation of which the radio spectrum is just a portion. Analysis of the light from the aurora reveals different colours and these colours are unique signatures of the elements that compose the upper atmosphere. From Wikipedia, the colours of the aurora have the following significance:
- Red: At its highest altitudes, excited atomic oxygen emits at 630 nm (red); low concentration of atoms and lower sensitivity of eyes at this wavelength make this color visible only under more intense solar activity. The low number of oxygen atoms and their gradually diminishing concentration is responsible for the faint appearance of the top parts of the "curtains". Scarlet, crimson, and carmine are the most often-seen hues of red for the auroras.
- Green: At lower altitudes, the more frequent collisions suppress the 630 nm (red) mode: rather the 557.7 nm emission (green) dominates. A fairly high concentration of atomic oxygen and higher eye sensitivity in green make green auroras the most common. The excited molecular nitrogen (atomic nitrogen being rare due to the high stability of the N2 molecule) plays a role here, as it can transfer energy by collision to an oxygen atom, which then radiates it away at the green wavelength. (Red and green can also mix together to produce pink or yellow hues.) The rapid decrease of concentration of atomic oxygen below about 100 km is responsible for the abrupt-looking end of the lower edges of the curtains. Both the 557.7 and 630.0 nm wavelengths correspond to forbidden transitions of atomic oxygen, a slow mechanism responsible for the graduality (0.7 s and 107 s respectively) of flaring and fading.
- Blue: At yet lower altitudes, atomic oxygen is uncommon, and molecular nitrogen and ionized molecular nitrogen take over in producing visible light emission, radiating at a large number of wavelengths in both red and blue parts of the spectrum, with 428 nm (blue) being dominant. Blue and purple emissions, typically at the lower edges of the "curtains", show up at the highest levels of solar activity.[23] The molecular nitrogen transitions are much faster than the atomic oxygen ones.
- Ultraviolet: Ultraviolet radiation from auroras (within the optical window but not visible to virtually all[clarification needed] humans) has been observed with the requisite equipment. Ultraviolet auroras have also been seen on Mars,[24] Jupiter and Saturn.
- Infrared: Infrared radiation, in wavelengths that are within the optical window, is also part of many auroras.[24][25]
- Yellow and pink are a mix of red and green or blue. Other shades of red, as well as orange, may be seen on rare occasions; yellow-green is moderately common.[clarification needed] As red, green, and blue are the primary colors of additive synthesis of colors, in theory, practically any color might be possible, but the ones mentioned in this article comprise a virtually exhaustive list.
Effect on HAM Radio
The penultimate bullet above is intriguing. The radio spectrum goes from about 30 Hz to 300 GHz. 300 GHz is at the far end of the infrared range but also at the higher end of the regulated radio spectrum. It is only natural that the effects of the aurora will be evident in radio communications - both directly and from the effects on the upper atmosphere (where radio waves are refracted off the layers of the upper atmosphere). It is possible to hear radio emissions from extra-terrestrial aurora - for example the planet Jupiter's aurora interacts with the aurora of its moons. NASA runs an interesting high school level project to detect the aurora by radio - Project Jove - where one builds a radio telescope and sends the data back to NASA. For amateur radio operators, we would call these signals QRN (natural noise) and they can be detected around 29 MHz. Sometimes the terrestrial aurora results in something called auroral flutter and can wipe out HF communications. For amateur radio operators the effects of the aurora are evident all year round. This is one of many atmospheric challenges that operators face in the North and why the VE8 callsign prefix (along with VY1 and VY0) are so rare to hear.
Aurora for Yellowknife
The City of Yellowknife has built up a bit of a reputation as a tourist destination for aurora viewing. Astronomy North maintains a website and promotes the aurora. It is an outreach educational organization and a partnership with the Canadian Space Agency, University of Calgary (physics department), Government of the Northwest Territories and City of Yellowknife. On the Home Page of YARS' website there is a Canadian government space weather RSS feed.
- Auroral flutter an interesting phenomenon - KB6NU's Ham Radio Blog
- The Sound of the Aurora Borealis (Northern Lights) - YouTube
- Listening to Northern Lights - YouTube
- Hubble Captures Vivid Auroras in Jupiter’s Atmosphere | NASA
- Aurora Forecast - City of Yellowknife
- https://astronomynorth.ca/
- http://astronomynorth.com/aurora-forecast/



